Project Participants: Jason Baer, Jenna Aquino, Anneke van der Geer, Ashton Ballard, Rohwer Lab (SDSU); Yun Scholten and Will Barnes, Haas Lab (NIOZ)
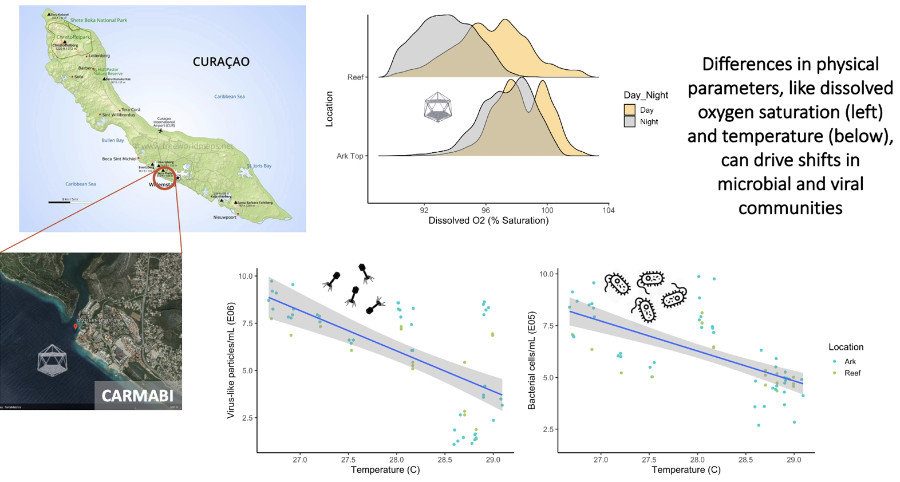
Microbialization is reinforced by changes in the chemistry of reef water (high DOC and low oxygen), making it challenging for corals to recover (Haas et al 2016). We hypothesize that by altering these chemical conditions, which we are doing by moving a coral reef community up into the water column where DOC is lower and O2 is higher, we will be able to break the DDAM feedback loop and reduce microbialization at the scale of that reef community.

As of March 2022, two Arks have been deployed in Curacao. These Arks contain limestone ARMS suitable for colonization by, and introduction of, viruses, microbes and invertebrates. Arks are positively buoyant and tethered to the seafloor to maximize O2 levels and flow.



Goal: Track assembly of Arks-associated communities to identify Ark structure and placement that maximizes (1) abundances of beneficial reef dwellers (e.g., invertebrates, viruses, some microbes), (2) coral health, (3) reef accretion, and (4) favorable water chemistry &l; dynamics.
Autonomous Reef Monitoring Structures (ARMS), originally developed as a tool to census and compare biodiversity across global environments (www.infinitediversity.org), are being used here to collect and move coral reef biodiversity in order to build a reef community. We are making ARMS out of limestone, a more natural material than the original PVC, and with differing complexities to study how three-dimensional space impacts the communities that recruit.

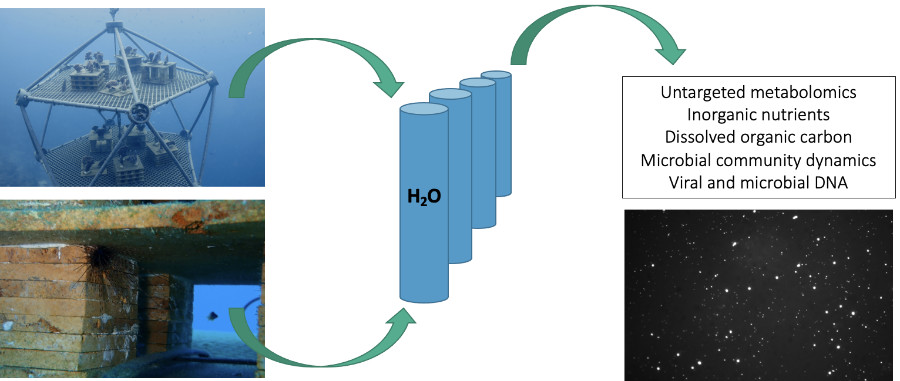
Water samples from ARMS and Arks are collected and processed to characterize the viruses, chemicals, nutrients, and microbes present in that seawater, each of which can give us an indication of ecosystem health. (Bottom right) An epifluorescence micrograph showing viral and bacterial communities in seawater: large white dots in the photo represent bacteria, while small white dots represent virus-like particles. These images are used to determine viral and bacterial abundances in seawater.
Master's student Jenna Aquino is monitoring the viral populations, microbial processes, chemical transformations, and oxygen concentrations inside the ARMS units as the communities within develop through time. Determining what processes different corals require from the suite of organisms they associate with will help us understand how to provide restored corals with the conditions they need to grow and survive. So far, it is clear that ARMS units with higher complexity (i.e., more surfaces) harbor different viral/microbial communities and invertebrate communities, which Jenna will focus on describing this year.
Master's students Yun Scholten and Will Barnes from the Haas lab tracked the newly deployed Arks through time. Yun and Will used a suite of advanced sampling techniques to study how the viruses and microbes are changing, and being changed by, the Arks environment and the surrounding seawater. By analyzing the chemicals in seawater produced and consumed by all the organisms that live in it, measuring concentrations of nutrients and organic carbon, and quantifying viral and microbial abundances, Will and Yun were able to show that the Arks reefs display a chemical and biological “signature” that is distinct from the surrounding seafloor or water column. The microbial communities on Arks, for example, display fewer signs of microbialization than the reef, with higher abundances of virulent viruses and microbial communities with lower biomass. From the data they collected from the Arks and the surrounding environment, Will and Yun were able to construct a detailed picture into how a coral reef community assembles at the smallest scales. This time series is ongoing, and as the Arks communities grow and change, we are continuing to track them through time.
The three-dimensional structure created by the Arks provides a refuge and a food source for reef grazers like fish and urchins. Master's student Ashton Ballard will be studying the grazing community that develops on Arks, including urchins, hermit crabs, and fish, to determine if these structures can be used to enhance grazing. Ashton is working on building Urchin Generating and Housing Structures, or (UrGHs) on Arks which will enhance urchin populations, and thus grazing processes, on Arks and the surrounding reefs. Ashton will also monitor custom settlement tiles on Arks and the surrounding areas to determine if the different coral species on the Arks can enhance larval production on the surrounding reef.

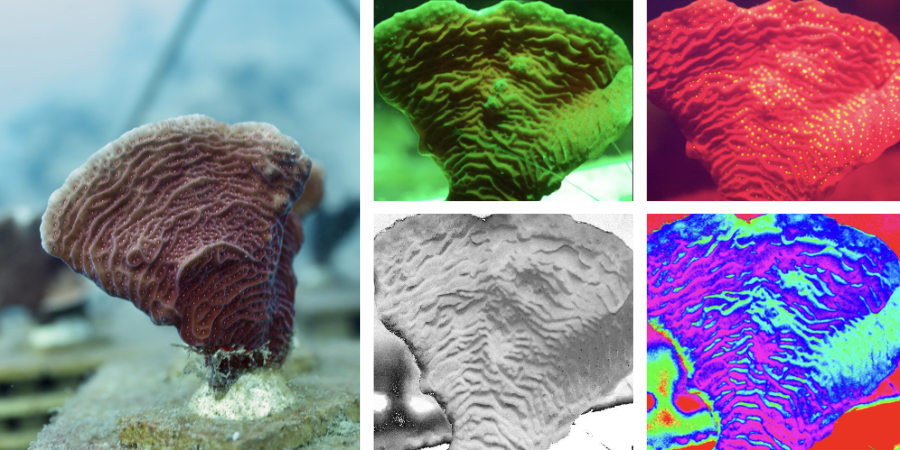
Whether it is possible to improve the health of a degraded coral reef community by moving it off the seafloor remains unknown. However, if this approach works, it will provide us with a means of conserving a reef's biodiversity in the midwater even as the surrounding natural reefs degrade. Master's student Anneke van der Geer is using a custom imaging device to measure the fluorescence of corals on Arks, which will tell us how efficient their photosynthetic processes are – a direct measure of a coral's health. Shown below is a sequence of images used to estimate the Fv/Fm ratio, a metric of photosystem health.
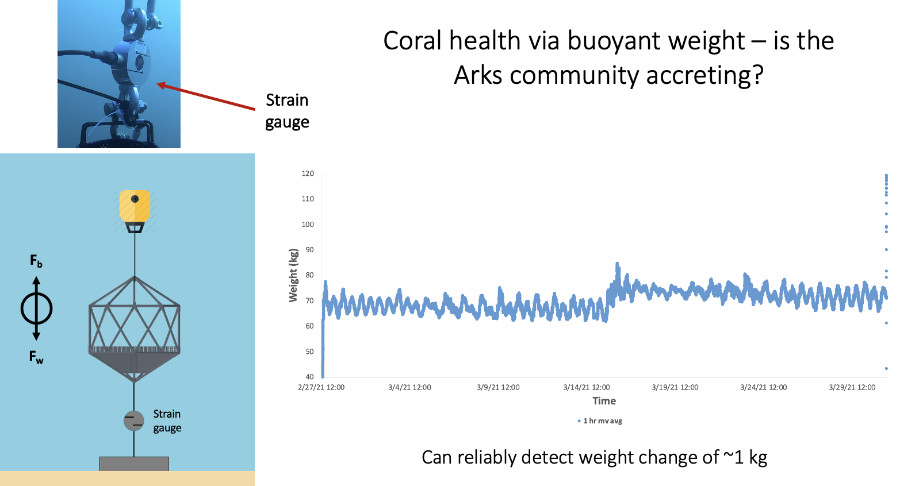
Using a novel submersible strain gauge system, we will weigh the Arks to determine if they are calcifying, a primary metric of coral health.
Our team moved a small cohort of corals (Agaricia agaricites) to the Arks as well as to a control site situated on the seafloor. In addition to closely monitoring their health (see #2 above), we are collecting data to characterize the physical environment on Arks, such as the flow speeds, oxygen levels, and light intensity relative to the seafloor. As coral communities develop on the Arks, we expect this physical environment to change as corals build three-dimensional structure. Jason Baer is using a submersible strain gauge to weigh the Arks over time to determine if the corals on them are growing by laying down a stony skeleton in a process called calcification.
Instruments deployed to measure physical parameters on Arks. (Left) The inert dye fluorescein is used to visualize flow inside the Arks. (Middle) Mark Little deploys an acoustic doppler current profiler to measure water flow speeds. (Right) Three probes are shown: an oxygen sensor, a multiprobe, and a current meter.

A reef community in the midwater experiences different physical conditions than a reef community on the neighboring seafloor. Higher dissolved oxygen concentrations, flow speeds, and light in the midwater provide an improved environment for coral growth relative to the surrounding degrading reef, and the results of these conditions on viral and microbial communities also impact coral health.